This content is intended for the general public
R&D PIPELINE
Blue Earth Diagnostics’ commitment is to develop and commercialise innovative radiopharmaceutical agents that have the potential to inform management decisions for patients with serious disease, such as cancer. These agents are designed to have a positive impact on patient care, with a focus on areas of significant unmet medical need. Here you can explore details of our research pipeline, arranged by product.
These products are undergoing clinical trials in the therapeutic areas listed and are not yet licensed.
|
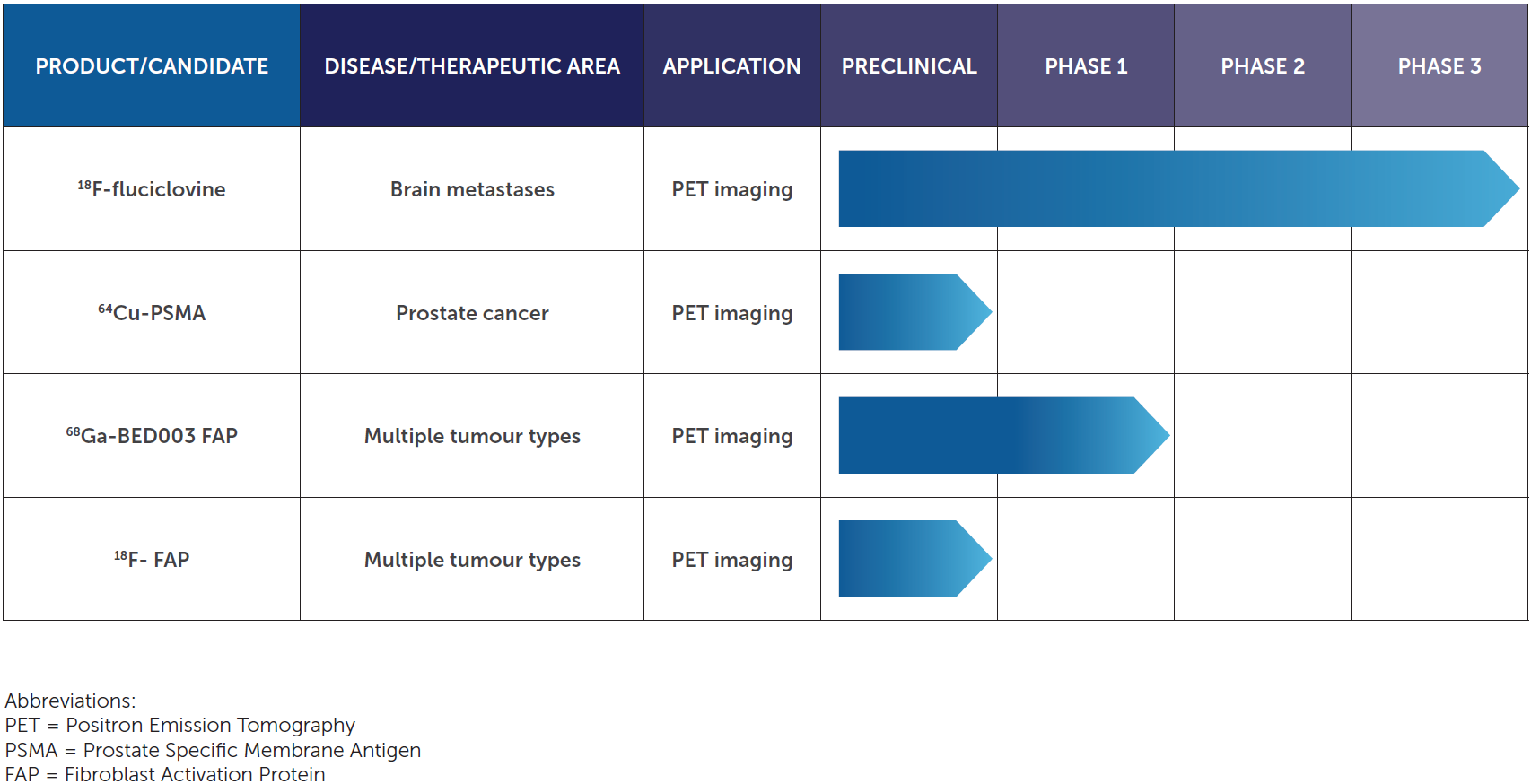
Product/Candidate
|
DISEASE/THERAPEUTIC AREA
|
Application
|
Preclinical
|
Phase 1
|
Phase 2
|
Phase 3
|
|
fluciclovine(18F) |
Brain metastases |
PET imaging |
|
|
|
|
|
FAP-targeted technology |
Multiple solid tumours |
PET imaging
|
|
|
Our sister company, Blue Earth Therapeutics, continues to investigate the use of radiopharmaceuticals in the therapy setting. For more information see:
https://www.blueearththerapeutics.com/pipeline
UKBED-N/A-2500024 April 2025
